When you’re streaming high-definition videos or holding 5G video conferences, have you ever wondered why the signal is so stable and latency so low? Beyond chip and antenna advancements, the real determinants of transmission quality are often the “invisible guardians”—electronic materials. In the era of high-frequency communication, copper clad laminate (CCL) resins face stringent requirements: low dielectric constant, low dissipation factor, high heat resistance, and low coefficient of thermal expansion (CTE). Traditional materials like polyimide or epoxy resins struggle to meet the demands of 5G/6G, while benzocyclobutene (BCB) derivatives are quietly emerging as a leading choice for high-performance electronic materials. Among them, 4-bromobenzocyclobutene (4-BrBCB) and 4-vinylbenzocyclobutene (4-VBCB) stand out as two key molecules—each with unique strengths, yet perfectly complementary when combined to drive CCL resin upgrades.

Spotlight on the Two Star Molecules: 4-Bromobenzocyclobutene vs. 4-Vinylbenzocyclobutene
The benzocyclobutene (BCB) family owes its outstanding performance to its unique structure: dielectric constant (Dk) typically 2.4–2.6, dissipation factor (Df) below 0.003 (at 1 GHz), glass transition temperature (Tg) exceeding 350°C, CTE in the 20–28 ppm/°C range (well-matched with silicon and copper substrates), and moisture absorption below 0.3%. It cures thermally without releasing volatile byproducts and offers volume resistivity above 10¹⁷ Ω·cm—making it ideal for high-frequency applications.
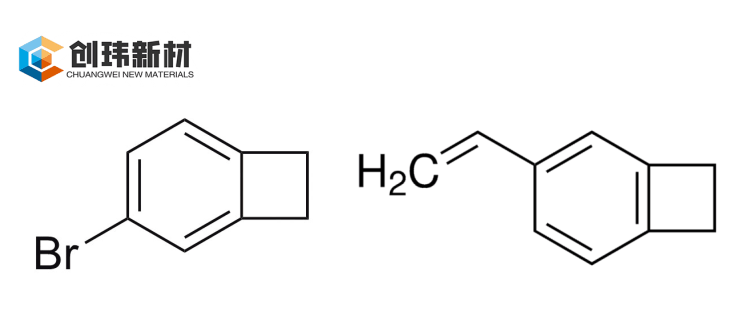
4-Vinylbenzocyclobutene (4-VBCB, CAS: 10365-98-7) acts like a “polymerization engine.” With its vinyl group and BCB ring, it enables direct free-radical, anionic, or coordination polymerization (e.g., rare-earth catalysis) to form linear prepolymers, followed by Diels-Alder ring-opening cross-linking of the BCB ring. This orthogonal curing mechanism allows precise control of the polymerization process—for example, copolymerization with styrene can adjust BCB content from 5–30 mol% to optimize Tg and mechanical properties. 4-VBCB exhibits excellent solubility (in toluene, THF, etc.), is suitable for spin-coating or casting, and cures at 200–250°C in minutes to hours.
In contrast, 4-bromobenzocyclobutene (4-BrBCB, CAS variant of 694-87-1) functions as a “chemical bridge.” The bromine atom serves as a highly reactive site for Grignard reactions, palladium-catalyzed coupling, or Suzuki reactions, making it a key intermediate for synthesizing advanced BCB monomers such as divinylsiloxane-bis-benzocyclobutene (DVS-bis-BCB). With purity typically >96% and good stability under inert conditions, 4-BrBCB is well-suited for large-scale resin production. Although it does not polymerize directly, it significantly simplifies the path from raw materials to final resins, with yields often exceeding 70%.
In summary, 4-VBCB excels in polymerization control and rapid prototyping, while 4-BrBCB shines in modification flexibility and cost efficiency. When used together, they achieve complementary performance: for instance, using 4-BrBCB to synthesize functionalized monomers and then 4-VBCB for copolymerization can increase cross-link density by approximately 25%.
Real-World Applications in CCL Resins: The Power of Synergy
In CCL resin development, the combination of 4-BrBCB and 4-VBCB directly addresses core challenges in high-frequency signal transmission. For high-frequency CCL insulation layers, traditional resins often have Dk > 3.0, resulting in severe signal attenuation. By using 4-BrBCB as the starting intermediate to synthesize DVS-bis-BCB resin and incorporating 4-VBCB to enhance the polymer network, Dk can be controlled to 2.2–2.5 and Df below 0.002 (at 1 GHz). This resin supports thin-film coating (1–30 μm thickness), adhesion >18 MPa, and CTE matching with copper foil, reducing interlayer stress and warpage. In 5G base stations or millimeter-wave devices, this translates to improved transmission efficiency and over 20% lower signal loss.
In 2.5D/3D packaging, chip stacking demands high planarity and interconnect isolation. 4-BrBCB enables one-step synthesis of DVS-bis-BCB precursors via silanization, delivering excellent thermal stability and low moisture absorption. Combined with 4-VBCB’s rapid curing (reducing process time by ~30%), the resin achieves passivation layers and via insulation at lower temperatures. Real-world examples show this combination in high-bandwidth memory (HBM) packaging yields optical loss <0.2 dB/cm and helium leak rates better than 3.0×10⁻⁴ Pa·cm³/s, meeting the stringent reliability standards of MEMS and RF modules.

Furthermore, in automotive electronics and high-speed optical communication (e.g., 800G/1.6T optical modules), the dual-molecule resin performs exceptionally well. 4-BrBCB’s modification potential allows incorporation of siloxane or amino groups, improving moisture resistance by ~40%; 4-VBCB ensures precise polymerization control, with Tg exceeding 400°C—ideal for extreme environments. Industry reports indicate that global packaging and testing leaders have adopted similar BCB resins to replace traditional polyphenylene oxide (PPO), while domestic manufacturers are achieving smooth transitions from lab to mass production through optimized synthesis routes.
Why This “Golden Duo” Deserves Attention
As semiconductor packaging evolves from 2.5D to 3D and high-frequency transmission advances toward 6G, material innovation has become the core engine of progress. The complementarity of 4-VBCB and 4-BrBCB is particularly valuable: the former offers precise polymerization control for rapid iteration and customized performance; the latter serves as an upstream intermediate that simplifies process chains and reduces overall costs. From a cost perspective, 4-BrBCB is more suitable for commercial CCL development, while pairing it with 4-VBCB balances performance and economics.
The localization trend further enhances their appeal. Chinese manufacturers have mastered high-purity synthesis and application, delivering more stable supply chains and faster response times, reducing reliance on overseas sources. When selecting materials, exploring this duo often yields more reliable and cost-effective solutions—especially in high-frequency applications where they help address soldering temperatures and signal interference challenges.
Outlook and Invitation
With the explosive growth of AI chips, next-generation communications, and automotive electronics, the BCB family—particularly 4-bromobenzocyclobutene and 4-vinylbenzocyclobutene—is poised to play an even greater role. In the future, these materials are expected to drive higher packaging density and propel the industry toward greater efficiency and sustainability.
References
- Martin SJ, Godschalx JP, Mills ME, et al. Development of a low-dielectric-constant polymer for the fabrication of integrated circuit interconnect. Adv Mater. 2000;12(23):1769-1778. https://www.sciencedirect.com/science/article/pii/S0167931796000615
- Wang X, Zhang Y, et al. Hydrolysis and condensation of a benzocyclobutene-functionalized precursor for the synthesis of high performance low-K polymers. RSC Adv. 2017;7(14):8430-8438. https://pubs.rsc.org/en/content/articlelanding/2017/ra/c7ra00141j
- Shaw JM, Laibinis PE, et al. Benzocyclobutene-based polymers for microelectronics. Chemical Innovation. 2001;31(12):12-18. https://pubsapp.acs.org/subscribe/archive/ci/31/i12/html/12so.html
- Dow Chemical Company. CYCLOTENE™ 4000 Series Advanced Electronics Resins: Processing Procedures. 2020. https://wiki.nanofab.ucsb.edu/w/images/a/ad/BCB-cyclotene-4000-revA.pdf
- The Dow Chemical Company. Engineering thermoplastics containing pendant benzocyclobutene groups. European Patent EP0251357B1. 1993. https://patents.google.com/patent/EP0251357B1/en
- Moore JA, et al. Recent Progress in Benzocyclobutene Related Polymers. IntechOpen. 2012. https://www.intechopen.com/chapters/38937
- Kim JH, et al. High-Density Patterned Array Bonding through Void-Free Divinyl Siloxane Bis-Benzocyclobutene Bonding Process. Polymers. 2021;13(21):3633. https://www.mdpi.com/2073-4360/13/21/3633
- Benzocyclobutene (BCB) Market Demand Space Broad, China’s Localization Process Advancing Continuously. New Thinking Industry Research. 2025-08-30. http://www.newsijie.com/chanye/huagong/jujiao/2025/0830/11357020.html
- Beijing Guangyin Polymer Technology Co., Ltd. “Move-in Ready” Attracts Major Players – Feicheng Chemical Industry Park Adds Another “Heavyweight”. Tencent News. 2025-06-17. https://news.qq.com/rain/a/20250617A06S1N00
- Daken Chem Limited. 3-(Chloromethyl)bicyclo[4.2.0]octa-1,3,5-triene, CAS NO.65886-91-1. https://www.dakenchem.com/product/cas-no-65886-91-1-3-chloromethyl-bicyclo-4-2-0-octa-1-3-5-triene





